Introduction to Biosimilars Industry
The biosimilars industry has emerged as a vital component of modern healthcare systems, providing cost-efficient alternatives to expensive biologic therapies. With the increasing prevalence of chronic diseases such as cancer, autoimmune disorders, and metabolic conditions, healthcare providers are under pressure to deliver effective treatments while controlling costs. Biosimilars, which closely resemble approved biologics in terms of safety and efficacy, are gaining strong acceptance among healthcare professionals and patients. Regulatory authorities across the globe are also supporting their adoption by establishing clear approval pathways, which is fostering innovation and competition. The growing need for affordable healthcare solutions continues to drive the expansion of biosimilars across global markets.
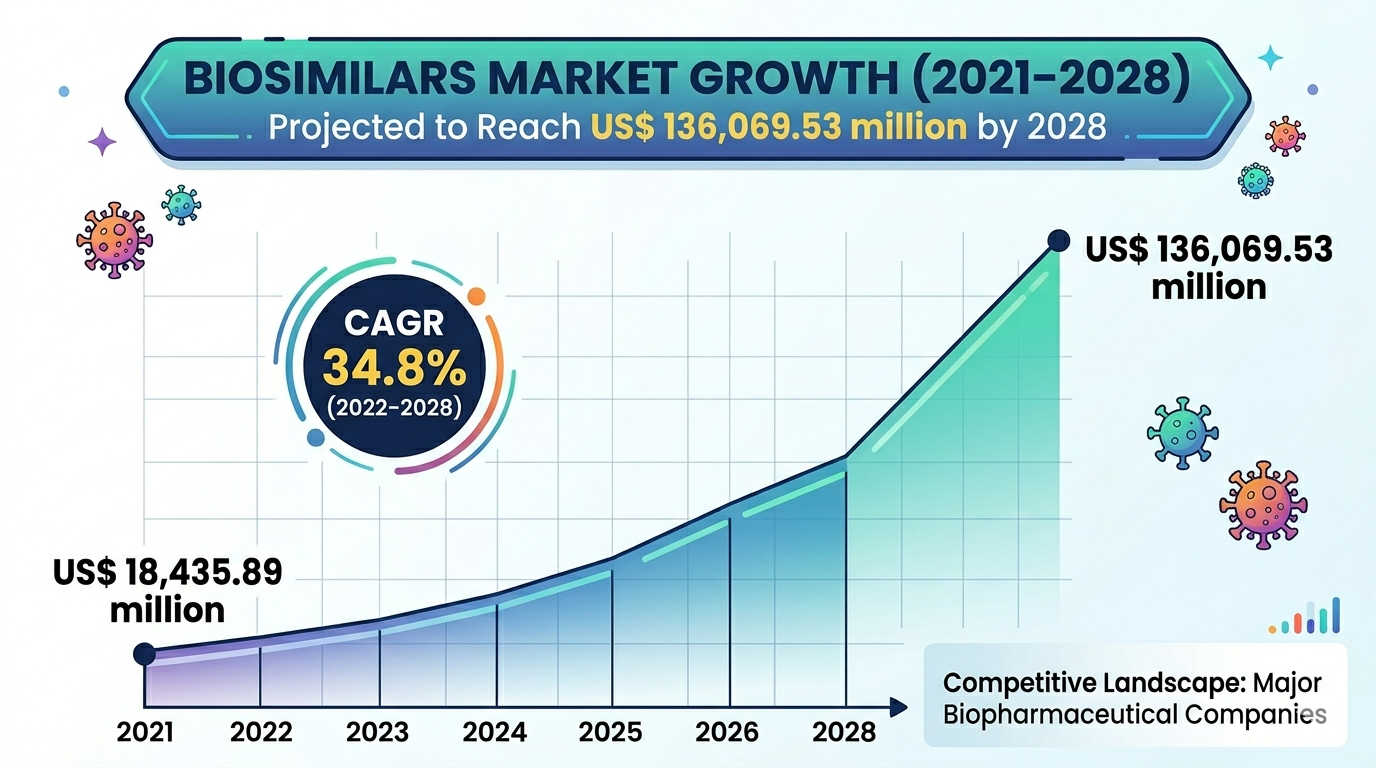
Market Overview and Expansion Dynamics
The Biosimilars Market is experiencing substantial expansion due to rising demand for biologic therapies and increasing cost-containment initiatives by healthcare systems. The industry is projected to grow at a significant CAGR during the forecast period, supported by a strong pipeline of biosimilar products and increasing regulatory approvals. Patent expirations of several blockbuster biologics are opening new opportunities for biosimilar manufacturers, allowing them to introduce cost-effective alternatives. Additionally, growing awareness among healthcare providers regarding the clinical effectiveness of biosimilars is contributing to their widespread adoption. Government initiatives promoting biosimilar usage are also playing a key role in driving industry growth.
Download Sample PDF Link:
https://www.theinsightpartners.com/sample/TIPHE100001246
Biosimilars Market Growth and Key Trends
The Biosimilars Market Growth is being driven by several evolving trends that are reshaping the industry landscape. One of the most significant factors is the increasing number of biologics losing patent exclusivity, which is creating a favorable environment for biosimilar development. Another important trend is the growing investment in research and development aimed at improving manufacturing efficiency and ensuring product quality. Strategic collaborations and partnerships among pharmaceutical companies are also accelerating product launches and expanding global reach. Furthermore, the increasing use of biosimilars in therapeutic areas such as oncology, immunology, and endocrinology is significantly contributing to overall growth.
Technological Advancements Driving Innovation
Technological advancements are playing a critical role in enhancing the development and production of biosimilars. Innovations in analytical characterization, cell line engineering, and bioprocessing technologies are ensuring high levels of similarity between biosimilars and reference biologics. Automation and digitalization in manufacturing processes are improving efficiency, reducing production costs, and shortening development timelines. Additionally, the use of artificial intelligence and machine learning in drug development is enabling better data analysis and optimization of production processes. These advancements are not only improving product quality but also strengthening the competitive positioning of biosimilar manufacturers.
Emerging Opportunities in Biosimilars Market
The Biosimilars Market offers significant growth opportunities, particularly in emerging economies where access to high-cost biologic therapies has traditionally been limited. Increasing healthcare investments and improving regulatory frameworks in regions such as Asia-Pacific, Latin America, and the Middle East are creating favorable conditions for market expansion. The rising focus on reducing healthcare expenditure is encouraging the adoption of biosimilars as cost-effective alternatives. Additionally, the growing emphasis on personalized medicine and targeted therapies is opening new avenues for innovation in biosimilar development. Partnerships between pharmaceutical companies and research organizations are further enhancing growth prospects.
Regulatory Landscape and Industry Challenges
The regulatory framework for biosimilars is evolving, with agencies such as the FDA and EMA establishing clear guidelines for product approval and commercialization. However, the development and approval process remains complex and requires extensive clinical studies to demonstrate similarity with reference biologics. High development costs and stringent regulatory requirements can pose challenges for new entrants. Additionally, concerns regarding interchangeability and physician acceptance may impact adoption rates in certain regions. Despite these challenges, ongoing efforts to harmonize regulatory standards and increase awareness are expected to support long-term industry growth.
Competitive Landscape and Strategic Developments
The Biosimilars Market is highly competitive, with numerous global and regional players focusing on innovation and strategic expansion. Companies are investing heavily in research and development to enhance product portfolios and improve manufacturing capabilities. Mergers, acquisitions, and collaborations are common strategies aimed at strengthening market presence and expanding geographic reach. The increasing demand for cost-effective biologic therapies is encouraging companies to develop advanced biosimilar products and improve operational efficiency. Continuous innovation and strategic initiatives are expected to drive competition and shape the future of the industry.
Key Players in Biosimilars Market
-
Pfizer Inc.
-
Novartis AG (Sandoz)
-
Amgen Inc.
-
Biocon Limited
-
Celltrion Inc.
-
Samsung Bioepis Co., Ltd.
-
Teva Pharmaceutical Industries Ltd.
-
Fresenius Kabi AG
-
Dr. Reddy’s Laboratories Ltd.
Future Outlook of Biosimilars Market
The future of the Biosimilars Market is expected to be highly promising, driven by continuous technological advancements and increasing demand for affordable biologic therapies. The growing number of patent expirations and expanding product pipelines will continue to create opportunities for market players. Advancements in biomanufacturing technologies and digital integration are expected to enhance efficiency and reduce development timelines. Additionally, increasing awareness among healthcare providers and patients regarding the benefits of biosimilars will further accelerate adoption. The industry is poised for sustained growth as it continues to play a crucial role in improving access to high-quality healthcare solutions.
Related Reports
Point of Care (PoC) Lipid Test Market
Clinical Laboratory Tests Market
About The Insight Partners
The Insight Partners delivers market intelligence and consulting services to help clients make informed decisions. The firm covers industries such as Aerospace and Defense, Automotive and Transportation, Semiconductor and Electronics, Biotechnology, Healthcare IT, Manufacturing, Medical Devices, Technology, Media, and Chemicals and Materials.
Contact Us
Email: sales@theinsightpartners.com
Website: www.theinsightpartners.com
Phone: +1-646-491-9876